|
11/17/2023 0 Comments Half life calculus examples 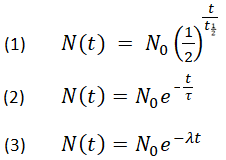
If we start with 10.0 g of carbon-14, the amount remaining after 5730 yr (1 half-life) will be 10.0 g × ½ 5.00 g. For example, carbon-14 has a half-life of 5730 yr. In nuclear chemistry, the half-life is the time needed for half of the radioactive atoms to decay. It can be determined experimentally for most practical situations since it depends on inner physical and chemical characteristics of a decaying substance.įor instance, half life of plutonium-239 is #24110# years, half life of caesium-135 is #2.3# million years, half life of radium-224 is only a few days. Half-life ( t ½) is the time required for a quantity to fall to half its value. So, all we need to know to find half life is the speed of a decay #K#. How to solve Half Life problems in calculus As far as solving half-life problems in calculus, perhaps an example would be useful. #1/2 = e^(-K*T)# (take natural logarithm) If 100.0 g of carbon-14 decays until only 25.0 g of. Calculate the half-life of a radioactive substance whose disintegration constant happens to be 0. Half Life Formula One can describe exponential decay by any of the three formulas N (t) N0 N (t) N0 N (t) N0 Where, N0 refers to the initial quantity of the substance that will decay. So maybe you start with the following 'classic' decomposition reaction: Math Processing Error Since there is only one reactant, you know it is first order. A good example can be that the medical sciences refer to the half-life of drugs in the human body which of biological nature. Denote m(t) the remaining mass of Cobalt-60 at time tyears. One more slightly involved example is when you derive half lives from rate laws in kinetics (you may see this in AP Chemistry, or in Physical Chemistry). (a)Write down the applicable di erential equation, given that radioactive substances decay at a rate proportional to the remaining mass. The above represents an equation with #T# being an unknown. Complete the following half-life problems be sure to show your thinking when solving problems. 5.Cobalt-60 has a half life of 5.24 years. 
In an interval twice as long (2 T) the nucleus survives only with a 25 probability (half of 50), in an interval of three half-life periods (3 T) only with 12.5 (half of 25), and. This probability amounts to 50 for one half-life. If at time #t=T# there is only half of the initial amount that is left, we have an equation: It is possible to determine the probability that a single atomic nucleus will 'survive' during a given interval. Exponential growth and decay show up in a host of natural applications. Half life is the value of #t# when there will be left only half of what was in the beginning at #t=0#.Īt #t=0# the value of our function equals to One of the most prevalent applications of exponential functions involves growth and decay models. Obviously, this function is descending from some initial value at #t=0# down to zero as time increases towards infinity.įor example, this function can represent a radioactive decay of certain quantity of plutonium-239 and describes the amount of plutonium-239 left after a time period #t#. If the growth rate per year is 4 of the current population, how long will it take for the population to double Example (Half-Life): Cellium-314 decays at a. Where #K# is a positive number characterizing the speed of decay. In either case, it is more appropriate to report the time since the plant has died as approximately 19,000 years since these measurements are never completely precise.Exponential decay is usually represented by an exponential function of time with base #e# and a negative exponent increasing in absolute value as the time passes: These are examples that model exponential growth and exponential decay. 
5730c = \ln$ then an approximate value of 19,030 years is found instead. Formulas for half life and formulas for population growth. Dividing by 10 first (to isolate the exponential expression) and taking the logarithm of both sides gives 
To solve for $c$, notice that $c$ is in the exponent and so we need to Since the half life of Carbon 14 is 5730 years, this means that afterĥ730 years there will only be 5 micrograms of Carbon 14 left in the preserved plant: Lets suppose that a well-known substance decomposes in water into chloride and sodium ions according to the law of exponential. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
